
Thus, by looking at an IR absorption spectrum, a chemist can make some important determinations about a molecule’s chemical structure. Atoms of individual elements emit light at only specific wavelengths, producing a line spectrum rather than the continuous spectrum of all wavelengths produced by a hot object. Because each of these bonds is different, each will vibrate in a different way, and absorb IR radiation of different wavelengths. Energy levels, with classifications and uncertainties, have been compiled for the spectra of the neutral atom and all positive ions of potassium (Z19). This produces an absorption spectrum, which has dark lines in the same position as the bright lines in the emission spectrum of an element. For example, a chemist might learn from an IR spectrum that a molecule contains carbon-carbon single bonds, carbon-carbon double bonds, carbon-nitrogen single bonds, carbon-oxygen double bonds, to name but a few. By using special equipment in the lab, chemists can look at the IR absorption spectrum for a particular molecule, and can then use that spectrum to determine what types of chemical bonds are present in the molecule. A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum. However, when separated using a prism or diffraction grating, the. Each element has its unique line emission spectrum. The wavelength of radiations absorbed by the matter contributes to the missing wavelength, which leaves dark spaces in the bright continuous spectrum. 
When hydrogen gas is placed into a tube and electric current passed through it, the color of emitted light is pink. For observing the absorption spectrum, electromagnetic radiations are bombarded on a sample which absorbs radiation of certain wavelengths. The production of line spectra by the atoms of an element indicate that an atom can radiate only a certain amount of energy. Each element has a different atomic spectrum. This line spectrum is called an atomic spectrum when it originates from an atom in elemental form. X-ray photoemission spectroscopy (XPS) has been used. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. The spectrum appears in a series of lines called the line spectrum. 
Just as the energy required to excite an electron in a particular atom is fixed, the energy required to change the vibration of a particular chemical bond is also fixed. To measure the degree of charge transfer from potassium to graphite-derived bands. This type of radiation is usually not energetic enough to excite electrons, but it will cause the chemical bonds within molecules to vibrate in different ways. 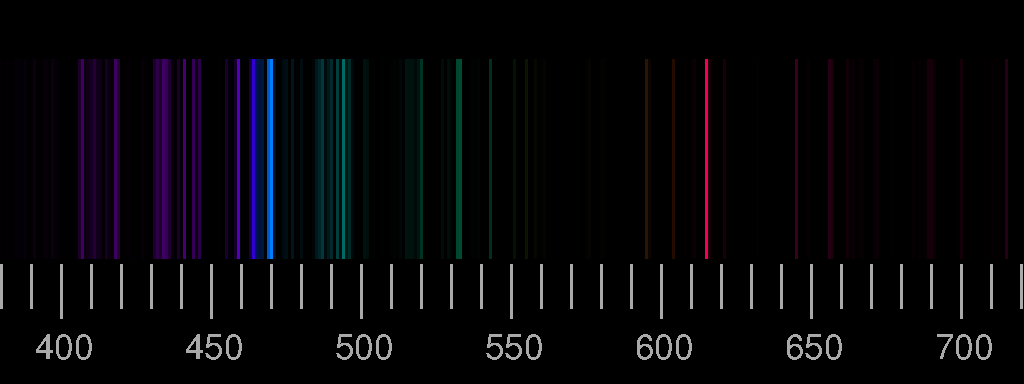
However, lower energy radiation in the infrared (IR) region of the spectrum can also produce changes within atoms and molecules. Potassium and cesium are frequently used as an ionization suppressor because of their low ionization energy. Although objects at high temperature emit a continuous spectrum of electromagnetic radiation (Figure 2.2.2), a different kind of spectrum is observed when pure samples of individual elements are heated. It may result from emission or absorption of light in a. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus.So far, we have been talking about electronic transitions, which occur when photons in the UV-visible range of the spectrum are absorbed by atoms. A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum. Below are the bright line spectra of four elements and the spectrum of an unknown gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
